Dalton (1766–1844) proposed that all matter in the universe is made of indestructible, unchangeable atoms-each type characterized by a constant mass-that undergo chemical reactions by joining with and separating from each other. Mann proposes the quark model (independently George Zweig does as well), which describes elementary particles that have no substructure (and therefore can't be split).Many consider 2008 the 200th anniversary of atomic theory, John Dalton’s momentous theory of the nature of matter. Glenn Seaborg, many discoveries of the transuranium elements, as well as many advances in nuclear medicine, including the development of I-131 for thyroid disease.Įlementary particles smaller than the atom Murray Gell Otto Hahn, regarded as the father of nuclear chemistry, discovers nuclear fission, along with Lise Meitner. This also made it possible to produce elements heavier than uranium in the lab. James Chadwick discovers the neutron component of the atomic nucleus, explaining the nuclear fission of uranium 235. He also integrated the Planck quantum theory, stating that when electrons change orbits they emit a quantum of discrete energy.Įrwin Shrodinger described how electrons move in wave form, and developed the Schrodinger equation which describes how the quantum state of a system changes with time. Neils Bohr developed the Bohr atomic model, with electrons travelling in orbits around the nucleus, and chemical properties being determined by how many electrons are in the outer orbits. He used a gold foil experiment, observing the scattering of alpha particles, and demonstrated for the first time the existence of the atomic nucleus. Structure of an atom developed (1909-1911)Įrnest Rutherford known as the father of nuclear physics, developed the theory for the structure of the atom.
This is known as the elementary charge, one of the fundamental physical constants. Robert Millikan measured the charge of a single electron. This was the basis of nuclear energy.Ĭharge of an electron measured (1908-1917) His quanta are now called photons by physicists.Īlbert Einstein, creates special and general theories of relativity, and hypothesizes about the particle nature of light. Max Planck introduced what would be known as quantum theory, stating that electromagnetic energy could only be emitted in quantized form. JJ Thomson discovers the electron, using properties of cathode rays. Pierre and Marie Curie theorized that radioactive particles cause atoms to break down, then releasing radiation that takes the form of energy and subatomic particles.

These rays weren't deflected by a magnetic field produced in the cathode ray tube. Wilhelm Rontgen discovered that certain chemicals glowed when exposed to cathode rays.

Sir William Crookes constructed a primitive cathode ray tube, which later became the basis for television. Also proposed that these atoms are spherical, and are in motion.
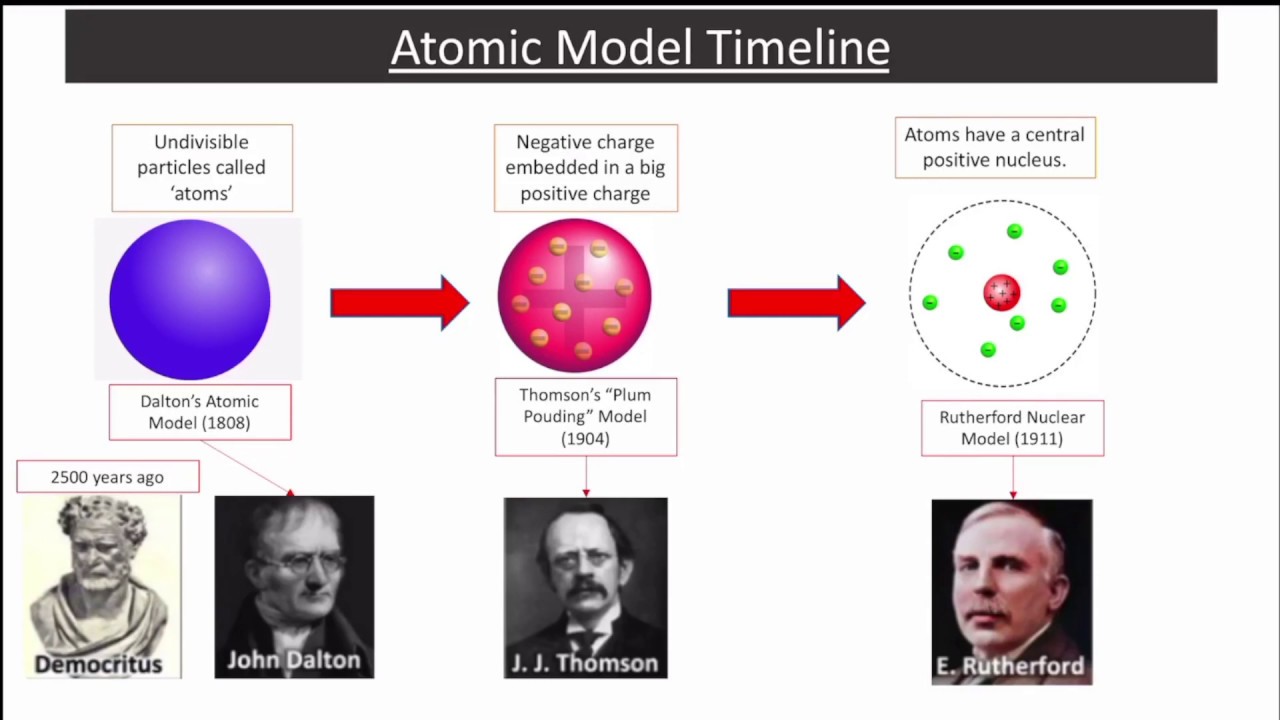
John Dalton, a British chemist and physicist, developed a theory that matter is simply composed of atoms of different weights and is combined in ratios by weight. Democritus, a Greek philosopher, co-originated the thought (with his teacher, Leucippus) that all matter is composed of indivisible elements.
